Tech
The Cảbon Chronicles: Unraveling the Chemical Conundrum
Cảbon – an unassuming element that holds the secrets to life as we know it. This basic building block of the universe, when combined with the right cocktails of other elements, creates wonders that range from the most delicate flower petals to the rugged blades of a bulldozer. For science enthusiasts, understanding the role of carbon is a key tenet of appreciating the vastness of our natural world.
In this comprehensive guide, we will take a deep dive into the multifaceted realm of carbon, which includes exploring its properties, significance, and influence on the broader picture of our planet. From the commonplace graphite in pencils to the enigmatic role of Cảbon in the cosmos, our expedition seeks to unravel the enigma of this elemental hero.
The Elemental Essence: What is Cảbon?
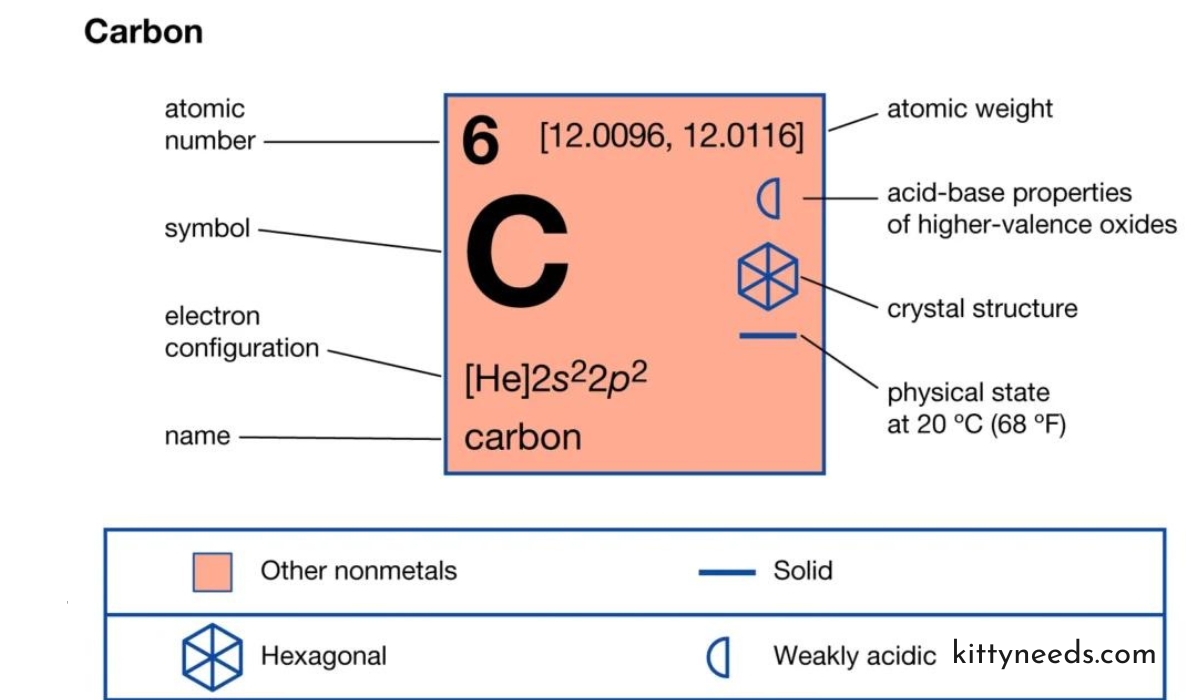
Cảbon is a chemical element that is represented by the symbol ‘C’ in the periodic table. With an atomic number of 6, it is classified as a nonmetal and is tetravalent – meaning it has the unique ability to form four covalent bonds with other atoms.
Despite its low relative abundance in Earth’s crust, hovering around a mere 0.025 percent, Cảbon ranks as the fourth most abundant element in the universe by mass, surpassed only by hydrogen, helium, and oxygen.
In essence, Cảbon is the unsung architect of the molecular world, playing a pivotal role in the formation of a vast spectrum of substances. It’s time to understand why this unassuming element commands such universal significance.
Tracing the Footprints of Carbon in Nature
The footprint of Cảbon can be found everywhere in the natural world, from the organic materials that compose the bodies of all living organisms to the inorganic compounds that shape geological formations.
Carbon’s Natural Allotropes: Diamond, Graphite, and More
Carbon presents itself in different structural forms, known as allotropes. The most celebrated of these are diamond and graphite.
Diamond is the crystal form of Cảbon and is renowned for its unparalleled hardness, making it an exceptional material for cutting tools and as a symbol of luxury. On the atomic level, diamond’s structure consists of a three-dimensional array of carbon atoms bonded in a tetrahedral arrangement.
Graphite, on the other hand, is a soft, black solid that serves as an excellent conductor of electricity, commonly used in pencil lead and certain industrial applications. Its atomic structure revolves around sheets of Cảbon arranged in a two-dimensional hexagonal lattice, aptly illustrating graphite’s malleability.
Fullerene, a relatively modern discovery, is a third form of Cảbon , featuring a hollow, cage-like structure. Buckminsterfullerene, the archetype of all fullerenes, resembles a soccer ball, with a network of twenty hexagons and twelve pentagons. Fullerenes have garnered commercial interest in nanotechnology and electronics.
The Chemical Symphony of Carbon Bonding
Carbon’s inclination towards catenation – the ability for atoms to form long chains via strong covalent bonds – is one of its most remarkable traits. This property underpins its ability to give rise to complex molecular structures and is the foundation of organic chemistry.
The covalent bonds that Cảbon forms are exceptionally strong, ensuring the stability of its compounds. This serves as a backbone for an exhaustive list of organic compounds that are central to life, such as proteins, carbohydrates, and nucleic acids.
Carbon and Sustainability
The role of carbon extends to environmental sustainability through Cảbon sequestration and its pivotal role in the carbon cycle. Understanding and harnessing carbon’s potential is crucial for maintaining the delicate equilibrium of our ecosystems.
Peering into the Nucleus: The Isotope Spectrum of Carbon
While the most common isotope of Cảbon , Cảbon-12, is the dominant variety constituting 98.93% of naturally occurring carbon, it’s important to acknowledge the other isotopes, particularly Carbon-14, with its implications in radiometric dating and other scientific applications.
The Carbon-14 Conundrum
Carbon-14, a radioactive isotope, has a half-life of approximately 5,730 years. This property makes it a pivotal tool in radiocarbon dating, which is used to determine the age of organic materials within a particular timeframe.
The use of Carbon-14 has provided invaluable insights into fields such as archaeology, paleontology, and environmental science, allowing for the establishment of a temporal framework for the natural and anthropogenic events that have shaped our planet’s history.
Carbon’s Crucial Role in Industry and Technology
The influence of carbon is felt keenly in the domains of industry and technology, its versatile properties making it a preferred building block for a myriad of applications.
Carbon as an Industrial Workhorse
From the carbon black used in the production of rubber to the carbon fibers lauded for their strength-to-weight ratio, carbon finds a multitude of vocational stations across the industrial landscape. Crucially, carbon-based materials are at the forefront of the push for sustainable and renewable energy solutions, with advancements in carbon capture and utilization (CCU) technologies.
Carbon in Technology:
The semiconductor industry leverages carbon’s conductivity and unique properties in the development of electronic and photonic devices. The rise of nanotechnology opens new frontiers, with carbon-based nanomaterials promising monumental breakthroughs in a wide range of fields, including medicine and energy storage.
The Carbon Cycle: Earth’s Natural Regulator
The carbon cycle is a robust and intricately woven network that governs the flux of carbon among the atmosphere, hydrosphere, geosphere, and biosphere. This natural system plays a fundamental role in regulating Earth’s climate by controlling the concentration of carbon dioxide in the atmosphere.
The Dance of the Carbon Atoms
Carbon moves through various forms and repositories during the carbon cycle, from its presence in the atmosphere as CO2 to its sequestration in rocks as carbonate minerals. The interplay between biotic and abiotic processes ensures a dynamic equilibrium that has persisted for aeons.
By understanding the complexities of the carbon cycle, we can appreciate the interconnectedness of Earth’s systems and the importance of maintaining a balanced carbon budget to preserve the planet for future generations.
Looking to the Stars: Carbon in the Cosmos
The prevalence of carbon in the cosmos is a testament to its affinity for stellar environments. From its role in the nucleosynthesis of elements within stars to the formation of carbonaceous chondrites, carbon bridges the gap between the terrestrial and the celestial.
A Cosmic Alchemist
Within the crucible of stars, carbon is synthesized through nuclear fusion processes, with carbon atoms serving as stepping stones for the creation of heavier elements. Supernovae and other high-energy phenomena scatter these elements across the cosmos, seeding future generations of stars and planets.
Implications for Astrobiology
The ubiquity of carbon in the universe has profound implications for the search for extraterrestrial life. Given carbon’s central role in biochemistry, the presence of carbon-based molecules beyond Earth could offer clues to the existence of life elsewhere in the cosmos.
Ethical Considerations and the Future of Carbon
With the increased awareness of climate change and the environmental impacts of carbon emissions, there is a growing emphasis on transitioning towards a carbon-neutral or even carbon-negative economy. Advancements in clean energy technologies and the development of novel materials are paving the way for a more sustainable future.
The Moral Imperative
The ethical dimensions of carbon’s role in the global climate crisis cannot be overstated. The imperative to act is clear, and the responsibility to future generations is profound. Addressing the challenges posed by carbon emissions requires a concerted effort, innovative solutions, and a commitment to stewardship.
Charting a New Course
From carbon capture and storage (CCS) to the emergence of carbon-negative materials, there is a rich tapestry of initiatives aimed at mitigating the impacts of carbon on the environment. These efforts not only seek to minimize harm but also to harness the potential of carbon in constructive and regenerative ways.
By embracing a holistic approach to carbon management, we have the opportunity to redefine our relationship with this elemental linchpin. The integration of sustainable practices, innovative technologies, and responsible policies can lead us towards a future where carbon is not a liability but an asset, elemental in both name and contribution.
YOU MAY ALSO LIKE
Pôde: Understanding the Nuances of the Portuguese Verb “Poder”
In Conclusion: Carbon, A Thread in the Tapestry of Life
The carbon atom, with its four valence electrons and unique bonding capabilities, weaves a remarkable tapestry of life on Earth and potentially beyond. Its significance is deeply entrenched in every domain of science and industry, from the microscopic world of biochemistry to the vast extremes of interstellar space.
The story of carbon is far from over. It continues to be written through the collaborative efforts of the scientific community, the ingenuity of researchers and engineers, and the global consensus on environmental stewardship. This is a tale of an ordinary element with extraordinary implications, a chemical conundrum that invites us to explore, understand, and ultimately, to act.
For science enthusiasts, the carbon chronicles offer an inexhaustible trove of knowledge waiting to be discovered. By peeling back the layers of this multifaceted element, we gain not only a deeper appreciation for the natural world but also insights that could shape the course of human progress for generations to come.
The carbon core is stable, the bonds unbreakable. What worlds would we discover if we dared to venture into the realm of carbon’s potential, and what legacies would we leave? The answers are as elemental as the question itself.
Frequently Asked Questions (FAQs) about Carbon
- What is the carbon cycle and why is it important?
- The carbon cycle is the process through which carbon atoms continually travel from the atmosphere into the Earth and then back into the atmosphere. This cycle is crucial because it regulates the Earth’s climate, helps to maintain the balance of carbon dioxide in the atmosphere, preventing extreme climate changes, and supports a wide range of life forms on our planet.
- How is carbon captured and stored?
- Carbon capture and storage (CCS) is a technology designed to collect carbon dioxide emissions produced from the use of fossil fuels in power generation and industrial processes, preventing it from entering the atmosphere. The captured carbon is then transported and stored underground in geological formations or used in various industrial processes.
- What role does carbon play in the search for extraterrestrial life?
- Due to carbon’s central role in biochemistry on Earth, scientists believe that carbon-based molecules could serve as indicators of life elsewhere in the universe. Because carbon is fundamental to DNA and other key components of life as we know it, its presence on other planets or moons could suggest the potential for extraterrestrial life.
- What are carbon-negative materials?
- Carbon-negative materials are materials that capture and store more carbon during their production than is emitted, effectively reducing the amount of carbon dioxide in the atmosphere. These materials, such as biochar, can be used in construction, agriculture, and various other fields to mitigate climate change.
- Why is transitioning to a carbon-neutral economy important?
- Transitioning to a carbon-neutral economy is crucial to combat climate change and minimize the negative impacts on the environment and human health. By reducing carbon emissions and enhancing carbon capture, it is possible to slow down global warming and work towards a more sustainable and healthier planet for future generations.

-

 Pets and Animals7 months ago
Pets and Animals7 months agoShovel Dog: Everything You Need to Know
-

 Pets and Animals7 months ago
Pets and Animals7 months agoHow To Trim Dog Nails: A Step-by-Step Guide for Painless Pups and Peace of Mind
-

 Pet Care Guides7 months ago
Pet Care Guides7 months agoGive a Loving Home: Adopt a Pet, Change a Life with Pet finder
-

 Pets and Animals7 months ago
Pets and Animals7 months agoHow Often Do You Take A Cat to the Vet? A Guide for Caring Owners
-
 Pets and Animals7 months ago
Pets and Animals7 months agoBubonic Plague case oregon cat: A Modern Tale of Medieval Disease
-

 Pet Care Guides7 months ago
Pet Care Guides7 months agoThe Definitive Guide to Rimadyl Carprofen Dogs: Uses, Dosage, and Safety
-

 Other7 months ago
Other7 months agoHow to Hello kitty drawing: A Step-by-Step Guide for Fans of All Ages
-

 Pet Care Guides7 months ago
Pet Care Guides7 months agoHere’s Why Nationwide Pet Insurance Should Be on Your Radar